Healthcare
Medical Devices
Precision Engineering Meets Autonomous Commerce
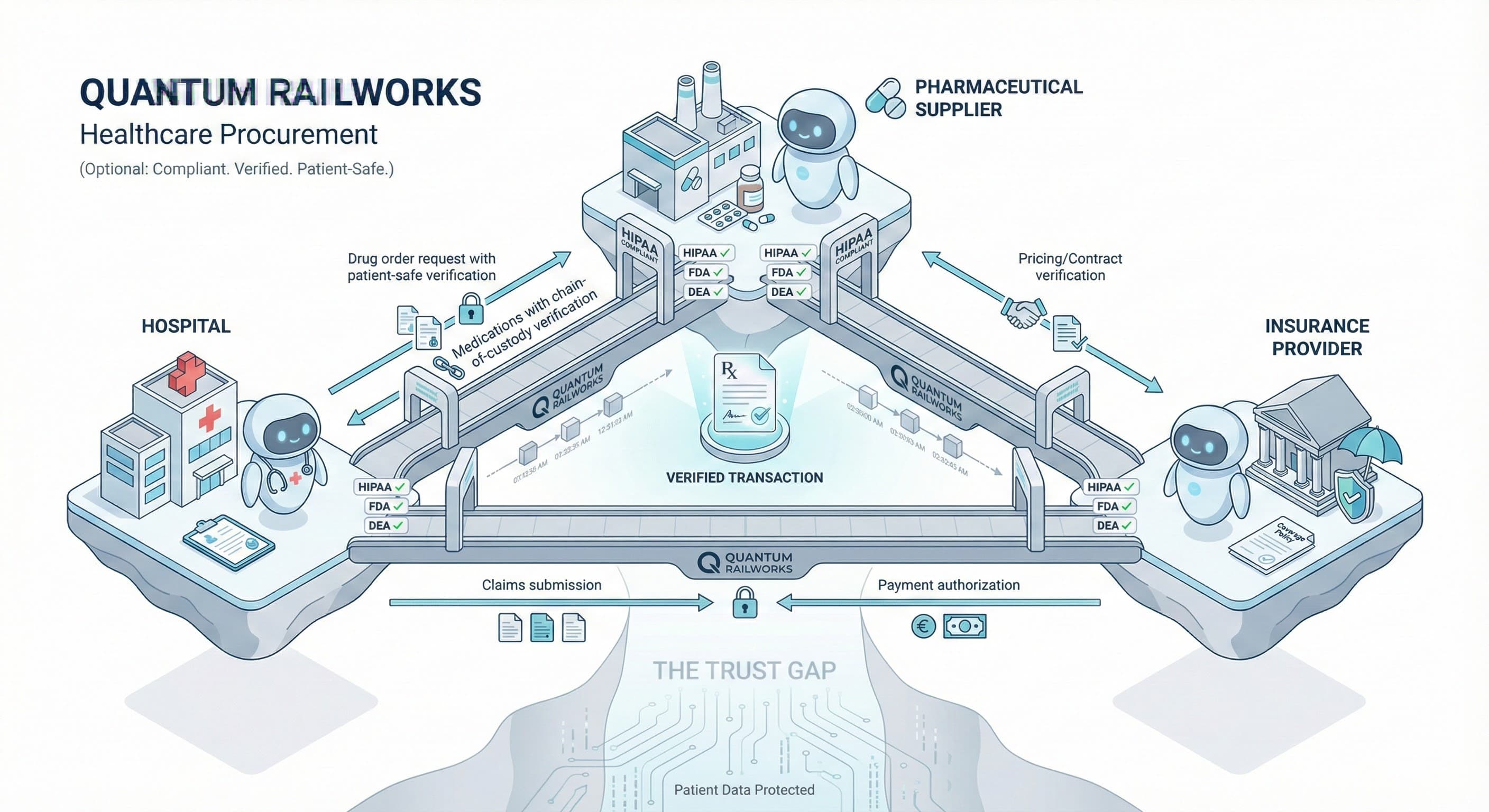
Medical device companies navigate complex ecosystems—contract manufacturers, component suppliers, distributors, GPOs, and healthcare providers. Quantum Railworks enables your AI agents to autonomously manage supplier relationships, distribution networks, and customer accounts while maintaining FDA compliance and quality standards.
The Challenge
Medical device operations demand precision across the entire value chain:
Supplier & Component Management Managing relationships with precision component suppliers, contract manufacturers, and sterilization providers requires rigorous quality control, supplier qualification, and capacity planning. Manual oversight can't scale with product portfolio complexity.
Distribution & Channel Complexity Navigating relationships with distributors, GPOs, IDNs, and direct accounts involves complex pricing structures, inventory consignment, and territory management. Manual coordination creates inefficiencies and channel conflict.
Service & Support Coordination Managing field service networks, third-party service providers, and depot repair facilities requires continuous coordination. Equipment uptime directly impacts patient care and customer satisfaction.
Regulatory & Quality Compliance Maintaining FDA QSR compliance, EU MDR conformity, and ISO certifications across global operations requires comprehensive supplier quality management, CAPA processes, and documentation control.
How Quantum Railworks Helps
Intelligent Supplier Quality Management
AI agents continuously monitor supplier performance, quality metrics, and compliance status. Automatically trigger audits, manage CAPAs, and qualify backup suppliers to ensure supply continuity and quality standards.
Autonomous Distribution Network Management
Deploy agents that optimize distributor relationships, manage GPO compliance, and coordinate territory coverage. Automatically resolve channel conflicts and ensure pricing consistency across your network.
Orchestrated Field Service Operations
Agents coordinate with field service engineers, third-party providers, and depot facilities. Automatically dispatch, schedule, and track service events while maintaining regulatory documentation.
Automated Compliance Monitoring
Continuously monitor supplier certifications, audit status, and regulatory changes. Agents ensure your supply chain maintains compliance and proactively address emerging requirements.
Key Capabilities
Agent Discovery
Find qualified suppliers, manufacturers, and service providers meeting specific certifications
Trust & Identity
Verify ISO certifications, FDA registrations, and quality system compliance in real-time
Smart Contracts
Automated supply agreements, distributor contracts, and service-level arrangements
Real-Time Messaging
Instant coordination across supply chain, distribution, and field service operations
Enterprise Security
Medical device-grade security meeting FDA and global regulatory requirements
Audit Trails
Complete documentation for FDA inspections, notified body audits, and quality reviews
Compliance & Security
Built for medical devices regulatory requirements.
Integration
Connects with your existing technology ecosystem.
ERP & PLM
- SAP S/4HANA, Oracle Cloud
- PTC Windchill, Siemens Teamcenter, Arena
Quality Management
- MasterControl, Greenlight Guru
- Veeva Vault QMS, ETQ Reliance
Field Service
- Salesforce Field Service, ServiceMax
- SAP FSM, IFS, Oracle Field Service
Distribution & CRM
- Salesforce Health Cloud, Veeva CRM
- SAP CRM, Oracle CX
- --
Ready to Transform Medical Devices?
### Optimize Your Medical Device Operations Leading device companies are deploying autonomous agents to improve supplier quality scores by 20%, reduce distribution costs by 15%, and accelerate field service response times. [Request a Demo](/contact) | [Read Medical Device Case Study](/insights/medical-device-case-study)